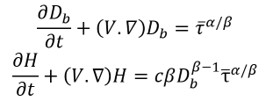Technical Challenge
When blood flows through a medical device, red blood cells may be subjected to damaging hemodynamic conditions typically not experienced in the human body, such as high shear stress. These high shear stresses may lead to hemolysis, which is the rupture of red blood cells that releases hemoglobin into surrounding fluid. The amount of free hemoglobin in blood indicates the degree of hemolysis.
To design medical devices that minimize the hemolysis rate, engineers can use computational models to predict both the patterns of blood flow and the risk of red blood cell damage. Utilizing computational models early in the development and design cycle allows clients to use this information in the next iteration of their product design, in an efficient process that reduces prototyping costs and device development time and optimizes product performance.
Veryst Solution
Veryst leveraged COMSOL Multiphysics to simulate the risk of hemolysis experienced in an FDA (U.S. Food and Drug Administration) benchmark study of a converging-diverging nozzle. The scenario considered for this case study involves injecting blood into a gradual contraction, followed by flow through a narrow channel and release into a rapid expansion. To achieve measurable hemolysis, the FDA benchmark uses a high flow rate of up to 6 L/min, which induces turbulent flow in this geometry. We simulated the turbulent flow using the SST (shear stress transport) turbulence model.
Note that this flow rate was chosen to induce experimentally observable hemolysis within an experiment lasting a few hours, but may be higher than biologically representative flow rates for some categories of passive devices.
We used two methods to model the hemolysis rate: a model in the Eulerian frame using transport of diluted species and a model in the Lagrangian frame using particle tracing.
These two methods target the same goal: to calculate the amount of hemoglobin released at the end of the device. Both methods give similar quantitative output, but the main difference involves the way hemolysis is averaged across the entire device.
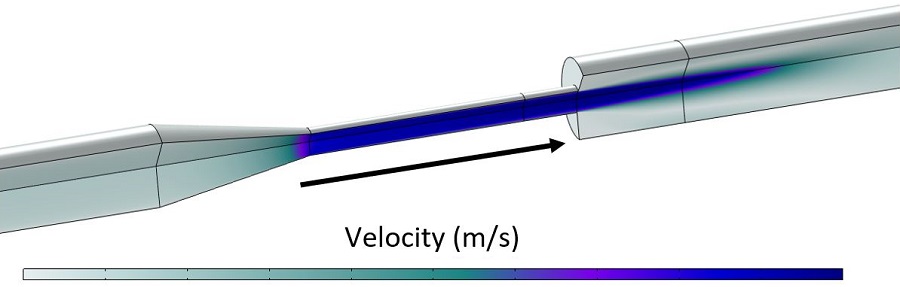
Figure 1 shows fluid velocity in the device, depicting the high velocity in the converging portion of the nozzle and a high velocity jet in the region with the rapid expansion. These velocities correspond to the 6 L/min case from the FDA benchmark. As noted previously, this flow rate was needed to generate experimentally observable hemolysis but may be physiologically high depending on the target device application. The simulation shown here is 2D axisymmetric, but we have also implemented this setup in 3D for other applications.
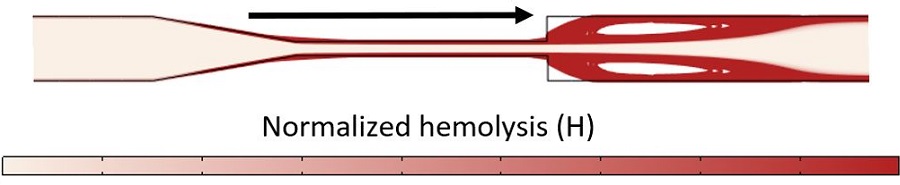
We present results from the Lagrangian solution in Figure 3, tracing the path of a blood cell initially near the inlet wall. Downstream in the rapid expansion, the damaged blood cells recirculate in a vortex generated by the jet. We simulated the device with the Lagrangian formulation to resolve potential recirculation regions better than is possible with the Eulerian formulation and to make it easier to highlight activity in the regions of greatest concern. When using a Lagrangian method, one must be careful to sample enough pathlines to capture all regions across the device to arrive at an averaged prediction for hemolysis, which is measurable in an experiment.
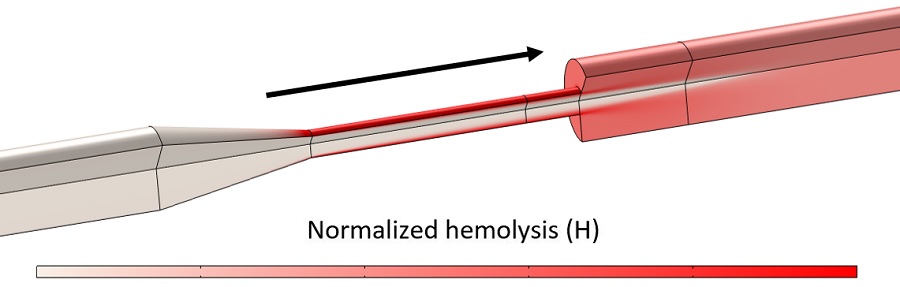
For the sake of brevity, we omitted details in generating Figures 2 and 3 and in translating the normalized hemolysis metrics shown here into absolute predictions of measured hemoglobin release. However, we want to emphasize that the amount of hemolysis in a single pass through the nozzle is quite small – less than one millionth of the total hemoglobin volume is released per pass – and the benchmark experiments ran flow in a closed loop for multiple hours to achieve measurable hemolysis from this nozzle. More quantitative measurements of hemoglobin loss and comparison to FDA benchmark data can be found in the COMSOL presentations linked at the bottom of this page.
Conclusion
Veryst modeled blood flow in a converging-diverging nozzle using COMSOL Multiphysics and estimated the relative risk of hemolysis due to elevated shear stress. Both the Eulerian and Lagrangian analyses identified the narrow region in the nozzle as the area with the most activity that could be associated with hemolysis.
These simulation methods and results are useful during the development of medical devices to ensure that the flow conditions do not induce hemolysis. We illustrated here the risk of blood damage by hemolysis, but it is also important to consider other failure pathways such as thrombosis (blood clotting) and how the risk of such failure modes increases if blood must pass repeatedly through a device.
Note that experimental measurements of hemolysis often involve passing blood through a device many times; the hemoglobin released on any single passthrough is generally small.
Veryst recommends comparing wall shear stresses with physiologically expected values to check for areas that pose a thrombosis risk when evaluating and designing medical and microfluidic devices. Quantifying the level of hemolysis in a medical device can also identify areas where there is risk of damage to red blood cells. Veryst can change the design near these risk areas to improve hemocompatibility.
About the Mathematical Modeling Used in this Case Study
The equations that describe the rate of hemolysis solve for two dependent variables: the mechanical dose (Db) and the ratio of the hemoglobin released into the surrounding fluid compared to the total hemoglobin in the red blood cell (H), following Grigioni et al (2005). The Eulerian method poses two advection equations in addition to the Navier-Stokes equations with velocity (V) and time (t) describing the transport of hemoglobin released by damaged blood cells in the flow. The hemolysis rate, described with source terms on the right side of the equation, has a power-law relationship with the shear stress (tau) experienced by the blood cells with experiments in the literature determining two coefficients (alpha, beta). There are several choices in the literature for values of alpha, beta, and c and we use the coefficients from Zhang et al. (2011).
The Lagrangian method integrates the equivalent source terms of the stress-based power-law relationship along pathlines through the fluid domain. We observe good agreement between the two methods. The Lagrangian method faces additional challenges in ensuring that flow pathlines of cells fill the entire geometry, whereas averaging the final hemolysis result is more straightforward with the Eulerian method.
___________________________
More details of Veryst’s approach can be found in the following presentations:
2021 COMSOL Days Keynote: https://www.comsol.com/video/keynote-blood-damage-modeling-of-fda-benchmark-nozzle
2020 COMSOL Conference: https://www.comsol.com/paper/blood-damage-modeling-of-fda-benchmark-nozzle-93171